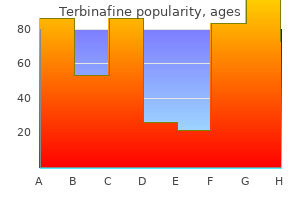
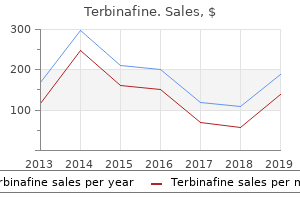
Terbinafine
"Generic terbinafine 250 mg line, antifungal probiotic".
By: K. Thorek, M.A.S., M.D.
Medical Instructor, Duquesne University College of Osteopathic Medicine
Administration of the second test could also be omitted when the test outcomes of the preliminary test meet the hearing requirement for that test antifungal in chinese buy terbinafine with a visa. Hearing necessities are: � � � � � First perceive a forced whispered voice fungus gnats damage plants discount terbinafine 250mg mastercard, in one ear antifungal treatment for grass discount terbinafine online amex, at not lower than 5 ft antifungal vitamins generic 250 mg terbinafine visa. When a hearing assist is used to meet the hearing qualification requirement, the hearing assist have to be used while driving. Health History and Physical Examination Health History Here are the hearing questions which might be asked within the well being history. Page 60 of 260 Physical Examination Examine the ears for: � � � Scarring of the tympanic membranes. Note and talk about irregular findings, including the impression on driving and certification. Required Tests the forced whisper test and audiometry are used to decide certification. These tests measure hearing loss utilizing the frequencies present in regular dialog. Administration of each tests is required solely when the preliminary test outcomes for each ears fail to meet the hearing requirement. When a driver who wears a hearing assist is unable to move a forced whisper test, referral to an audiologist, otolaryngologist, or hearing assist heart is required. When a hearing assist is used to qualify, the hearing assist have to be worn while driving. The testing area should be free from noise that might intrude with a valid test. From the measured 5-foot distance from the proper ear, exhale totally after which whisper a sequence of words, numbers, or letters. Complete the forced whisper test for each ears, whether or not the preliminary test result meets the hearing requirement. Page sixty one of 260 Audiometric Test the hearing qualification requirement for the Audiometric test: � � � Has an average hearing loss (common of test outcomes for 500 hertz (Hz), 1,000 Hz, and a couple of,000 Hz). The hearing requirement for an audiometric test is predicated on hearing loss solely on the 500 Hz, 1,000 Hz, and a couple of,000 Hz frequencies which might be typical of regular dialog. The area chosen for testing should be free from noise that might intrude with a valid test. Hearing assist When a hearing assist is to be worn during audiometric testing, an audiologist or hearing assist heart should carry out the test utilizing applicable audiometric gear. Additional Evaluation and/or Ancillary Tests Ear trauma and otic disease can adversely impression hearing and/or steadiness and intrude with safe driving and efficiency of associated duties. When findings are inconclusive relating to medical fitness for obligation, ancillary tests and/or additional analysis by a specialist, often an audiologist or otolaryngologist, could also be required to get hold of sufficient medical info to decide certification standing. The driver who makes use of a hearing assist to qualify should wear a hearing assist while driving. The examiner should advise the driver to carry a spare power supply for the hearing assist. Page 62 of 260 Advisory Criteria/Guidance Otic Preparations Determine if the therapy is having the specified effect of preserving hearing, reducing inflammatory problems causing ache, and/or controlling dizziness causing loss of steadiness. Determine if the therapy has any effects and/or unwanted effects that intrude with safe driving (e. Categories embody: Anti-acute Benign Positional Vertigo Classifications of brokers used to deal with acute vertigo embody: � � � � Antihistaminic antiemetics. Classification of oral medicine used to deal with infections and irritation of the center ear (otitis media) embody: � � Antibiotics. The Conference on Neurological Disorders and Commercial Drivers report recommends that the driver could also be certified after completing no less than 2 months symptom free with a analysis of: � � Benign positional vertigo. To evaluation the Conference of Neurological Disorders and Commercial Drivers report, go to:. Hypertension Americans With Hypertension According to the Third National Health and Nutrition Examination Survey, 29% of all U.
Bleeding related to eptifibatide concentrating on greater threat sufferers with acute coronary syndromes: incidence and multivariate threat components antifungal nail medication purchase terbinafine discount. Contraindicated medicine use in dialysis sufferers present process percutaneous coronary intervention fungus doctor order 250 mg terbinafine with visa. If the affected person undergoes percutaneous coronary intervention while receiving eptifibatide antifungal liquid review order terbinafine 250mg online, the infusion ought to be continued as much as fungus gnats how to get rid of naturally cheap terbinafine american express hospital discharge (max ninety six h) or for as much as 18�24 h after the process, whichever comes first. Infusion ought to be continued till hospital discharge or for as much as 18�24 h, whichever comes first. A part I examine of eribulin mesylate (E7389), a mechanistically novel inhibitor of microtubule dynamics, in sufferers with advanced stable malignancies. The primary antimitotic mechanism of motion of the synthetic halichondrin E7389 is suppression of microtubule growth. Eribulin binds at microtubule ends to a single web site on tubulin to suppress dynamic instability. Phase I examine of eribulin mesylate administered once each 21 days in sufferers with advanced stable tumors. Eribulin induces irreversible mitotic blockade: implications of cell-based pharmacodynamics for in vivo efficacy under intermittent dosing circumstances. Pharmacokinetics of free ertapenem in critically ill septic sufferers: intermittent versus steady infusion. Pharmacokinetics of once-every day dosing of ertapenem in critically ill sufferers with severe sepsis. Pharmacokinetics of ertapenem in critically ill sufferers with acute renal failure present process prolonged every day dialysis. Ertapenem in critically ill sufferers with early-onset ventilator-related pneumonia: pharmacokinetics with particular consideration of free-drug concentration. Underdosing of ertapenem in critically ill sufferers with pneumonia confirmed by Monte Carlo simulations [letter]. Comparative pharmacokinetics and pharmacodynamic target attainment of ertapenem in normal-weight, overweight, and very overweight adults. Pharmacokinetics of ertapenem following intravenous and subcutaneous infusions in sufferers. Probability of pharmacodynamic target attainment with normal and extended-infusion antibiotic regimens for empiric therapy in adults with hospital-acquired pneumonia. Pharmacokinetics of ertapenem in sufferers with varying levels of renal insufficiency and in sufferers on hemodialysis. Comparative disposition of [14C]ertapenem, a novel carbapenem antibiotic in rat, monkey and man. Ethacrynic acid: evaluation of saluretic and diuretic efficiency in sufferers with severe persistent renal failure. Effects of ethacrynic acid (a new saluretic agent) on renal diluting and concentrating mechanisms: proof for web site of motion in the Loop of Henle. Prolonged administration of ethacrynic acid in sufferers with persistent renal illness. Ethacrynic acid: diuretic property coupled to response with sulfhydryl teams in renal cells. Transient and permanent deafness following treatment with ethacrynic acid in renal failure. Intravenously given ethacrynic acid gastrointestinal bleeding: a finding resulting from comprehensive drug surveillance. Ethambutol optimum medical dose and susceptibility breakpoint identification by use of a novel pharmacokinetic-pharmacodynamic model of disseminated Mycobacterium avium. New susceptibility breakpoints for first-line antituberculosis medication based on antimicrobial pharmacokinetic/ pharmacodynamic science and population pharmacokinetic variability. Clearance calculations in hemodialysis: software to blood, plasma, and dialysate measurements for ethambutol. Pharmacokinetics of ethambutol under fasting circumstances, with food, and with antacids.
Adult Hypertension Diovan (valsartan) has been evaluated for safety in more than four antifungal tube buy terbinafine 250 mg with amex,000 patients fungus yellow mulch terbinafine 250mg overnight delivery, together with over four hundred treated for over 6 months diabet x antifungal skin treatment order terbinafine 250 mg mastercard, and more than one hundred sixty for over 1 year antifungal and antibacterial cream discount terbinafine 250mg line. Adverse reactions have usually been mild and transient in nature and have solely infrequently required discontinuation of therapy. The overall frequency of adverse reactions was neither dose-related nor related to gender, age, race, or routine. The commonest reasons for discontinuation of therapy with Diovan had been headache and dizziness. The adverse reactions that occurred in placebo-controlled clinical trials in a minimum of 1% of patients treated with Diovan and at the next incidence in valsartan (n=2,316) than placebo (n=888) patients included viral an infection (3% vs. An improve within the incidence of dizziness was noticed in patients treated with Diovan 320 mg (8%) in comparison with 10 to one hundred sixty mg (2% to four%). Pediatric Hypertension Diovan has been evaluated for safety in over four hundred pediatric patients aged 6 to 17 years and more than one hundred sixty pediatric patients aged 6 months to 5 years. No related variations had been recognized between the adverse experience profile for pediatric patients aged 6 to sixteen years and that previously reported for adult patients. Headache and hyperkalemia had been the most typical adverse events suspected to be examine drug-related in older youngsters (6 to 17 years old) and younger youngsters (6 months to 5 years old), respectively. Neurocognitive and developmental assessment of pediatric patients aged 6 to sixteen years revealed no overall clinically related adverse impact after remedy with Diovan for as much as 1 year. In a examine (n=ninety) of pediatric patients (1 to 5 years), two deaths and three circumstances of on-remedy transaminase elevations had been seen within the one-year open-label extension part. These 5 events occurred in a examine population during which patients incessantly had important co-morbidities. In a second examine of 6-months duration in seventy five youngsters aged 1 to 5 years, there have been no deaths; one case of marked liver transaminase elevations occurred following 6 months of remedy. The commonest adverse reaction in youngsters lower than 6 years old was hyperkalemia. Heart Failure In the Valsartan Heart Failure Trial, evaluating valsartan in total daily doses as much as 320 mg (n=2,506) to placebo (n=2,494), 10% of valsartan patients discontinued for adverse reactions vs. The desk exhibits adverse reactions in double-blind brief-term coronary heart failure trials, together with the first four months of the Valsartan Heart Failure Trial, with an incidence of a minimum of 2% that had been more frequent in valsartan-treated patients than in placebo-treated patients. All patients acquired commonplace drug therapy for coronary heart failure, incessantly as a number of medications, which may embrace diuretics, digitalis, beta-blockers. Valsartan (n=3,282) 17% 7% 5% 3% 3% 3% 2% 2% 2% Placebo (n=2,740) 9% 2% four% 2% 2% 2% 1% 1% 1% Dizziness Hypotension Diarrhea Arthralgia Fatigue Back Pain Dizziness, postural Hyperkalemia Hypotension, postural Discontinuations occurred in zero. Other adverse reactions with an incidence greater than 1% and greater than placebo included headache, nausea, renal impairment, syncope, blurred vision, upper belly pain and vertigo. In submit-myocardial infarction patients, doubling of serum creatinine was noticed in four. Digestive: Elevated liver enzymes and very rare reports of hepatitis Musculoskeletal: Rhabdomyolysis Renal: Impaired renal perform, renal failure Dermatologic: Alopecia, bullous dermatitis Blood and Lymphatic: Thrombocytopenia Vascular: Vasculitis 7 7. If co-medication is considered essential, monitoring of serum potassium is advisable. Published reports embrace circumstances of anhydramnios and oligohydramnios in pregnant ladies treated with valsartan (see Clinical Considerations). When being pregnant is detected, think about different drug remedy and discontinue Diovan as quickly as possible. Clinical Considerations Disease-related maternal and/or embryo/fetal risk Hypertension in being pregnant increases the maternal risk for pre-eclampsia, gestational diabetes, untimely supply, and supply issues (e. Hypertension increases the fetal risk for intrauterine development restriction and intrauterine death. Pregnant ladies with hypertension must be fastidiously monitored and managed accordingly. Fetal/Neonatal Adverse Reactions Oligohydramnios in pregnant ladies who use medicine affecting the renin-angiotensin system within the second and third trimesters of being pregnant can result in the following: reduced fetal renal perform leading to anuria and renal failure, fetal lung hypoplasia, skeletal deformations, together with cranium hypoplasia, hypotension and death. In patients taking Diovan during being pregnant, perform serial ultrasound examinations to assess the intra-amniotic setting. Closely observe neonates with histories of in utero publicity to Diovan for hypotension, oliguria, and hyperkalemia. In neonates with a history of in utero publicity to Diovan, if oliguria or hypotension happens, support blood stress and renal perfusion.
Buy generic terbinafine 250 mg on line. ഇങ്ങനെ ചെയ്താൽ പേരക്ക മരം തഴച്ചുവളരും | How To Protect Guava Tree From Fungus Disease.